Clinical trials can be expensive to run. Due to the high cost of running one, Big Pharma conduct most of the clinical trials. In a couple of instances, Big Pharma has been in the spotlight for influencing the results of clinical trials. A negative influence on clinical trial results and outcomes may be dangerous to the population.
The scope of Clinical trial sponsors has actively changed over a couple of years to include independent organizations and professionals, following designs and protocols agreed upon by all parties and approved by ethical review boards.
Long-term phase 3 trials are vital to sponsors and aimed at providing an adequate basis for a submission for marketing approval. These studies have a long duration and are likely to show long-term or rare side effects. Studies in phase 3 are conducted across a large patient population and should include a population representative of the intended target population that will use the drug.
Hence, phase 3 trials can be multicenter, occurring in different countries to provide diversity on the population target size. Here are some of the Sponsors of Phase 3 multicenter clinical trials that are ongoing in Kenya.
#Data Visualization: Primary Sponsors of on going Phase 3 Clinical Trials in Kenya
## Data ordered in reference to Population Target size
plot1 <- ggplot(data = p3,
aes((y = reorder(Primary_sponsor, -Target_size)), x = Target_size, fill = Target_size))+
geom_bar(stat = "identity", show.legend = FALSE)+
labs (title = "Phase 3 Clinical Trials in Kenya",
subtitle = "In 01-01-2022 to 31-10-2022",
y = "Primary Sponsors",
x = "Population Target size")+
theme_few()
#Data Source: trialsearch.who.int
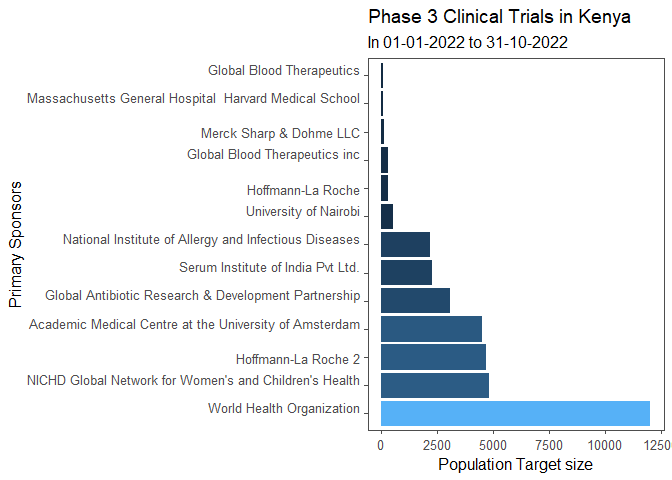
- World Health Organization
- Title: Low dose magnesium sulphate for treatment of seizures in pregnancy
- Population Target size: 12000
- Condition/Disease: Pre-Eclampsia
- Drug: Magnesium Sulphate
- Estimated completion date: 2023
- Global Network for Womens’ and Childrens’ Health
- Title: Prevention of Iron Deficiency Anemia Post-delivery (PRIORITY)
- Population Target size: 4800
- Condition/Disease: Postpartum anemia
- Drug: Iv iron infusion, Oral tablets
- Estimated completion date: December 1, 2025
- Hoffman La Roche
- Title: Phase-iii study to determine safety and efficacy of Giredestrant compared with physicians choice in patients with Estrogen Receptor Positive, HER2 negative early breast cancer
- Condition/Disease: Breast Cancer
- Drug: Giredestrant, Endocrine Therapy of Physician’s Choice, LHRH Agonist
- Estimated completion date: December 19, 2025
- Academic Medical Centre at the University of Amsterdam
- Title: Evaluation of a new and simple diagnostic test for malaria
- Population Target size: 4700
- Condition/Disease: Malaria
- Drug: Rapid Diagnostic Test
- Estimated completion date: 2023
- Global Antibiotic Research and Development Partnership
- Title: NeoSep1: a study to determine the ranking of existing and new antibiotics combinations to treat newborn babies who are in hospital with severe sepsis
- Population Target size: 3060
- Condition/Disease: Neonatal sepsis
- Drug: Amikacin, Flomoxef and Fosfomycin
- Estimated completion date: 2023